The gross heat of combustion value - also known as energy value or calorific value - is used to quantify the energy content of a cable in case of a fire. Cableizer calculates the value automatically for each cable.
Posted 2018-03-22
Categories:
New feature
, Theory
, Validation
The standard gross heat of combustion is the energy liberated when a substance undergoes complete combustion with oxygen at standard conditions (25°C and 1 bar). In thermodynamical terms it is the negative enthalpy change for the combustion reaction. The chemical reaction is typically a hydrocarbon or other organic molecule reacting with oxygen to form carbon dioxide and water and release heat. It can be calculated from the standard enthalpy of formation (ΔHf°) of the substances involved in the reaction, given as tabulated values. The gross heat of combustion is measured in a calibrated oxygen bomb calorimeter. It is usually utilized to quantify the performance of a fuel in a combustion system.
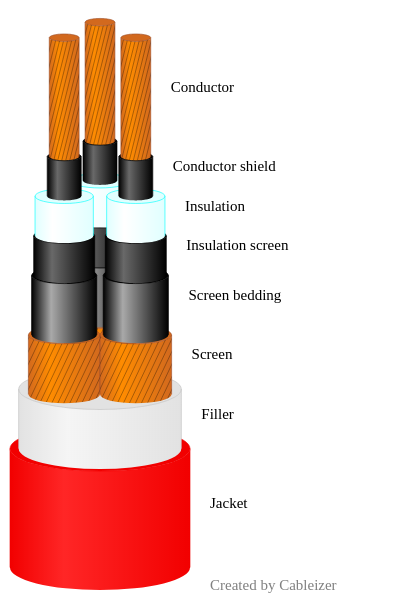
The heat energy content is calculated as a product of the mass per meter and the heat of combustion value per kg for all non-metallic materials. Cableizer separately calculates the heat energy content for the insulation, screen bedding, screen serving, filler, and jacket of your cable.
First, the mass of each cable element is calculated. This looks like the following in the cable editor:
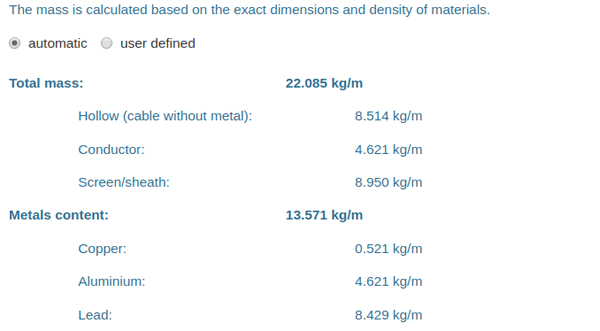  |
Second, the mass of each non-metallic element is multiplied with the specific heat of combustion value. All resulting values are then added up and the total heat energy content is presented to the user in MJ/m as well as kWh/m. This looks like the following in the cable editor:
 |
The combustion heat of cables and wires with nominal voltages up to 1000 V is usually taken from standard tables. For example, in the German speaking countries, the publication from VdS, a leading independent testing institution for fire protection and security is taken as a reference. Please refer to the English version of the leaflet for the Calculation of Fire Loads called Combustion Heat of Insulating Materials of Cables and Wires on their website.
There is a long table with typical values for standardized design of low voltage cables and wires beginning at a simple NYM 1x1,5mm² over NYY or NHXHX 5x2,5mm² up to NYCY or NHXCHX 4x150/70mm². They also list combustion heat of wires for telecommunication and data processing systems between 2x2x0,6mm² and 80x2x0,8mm².
But when it comes to cables with nominal voltages exceeding 1000 V there is only very little typical data available. Some medium voltage cable designs are more or less standardized in the Harmonized Documents HD 620, but tables with typical values are scarce. The VdS leaflet contains only 5 typical cables:
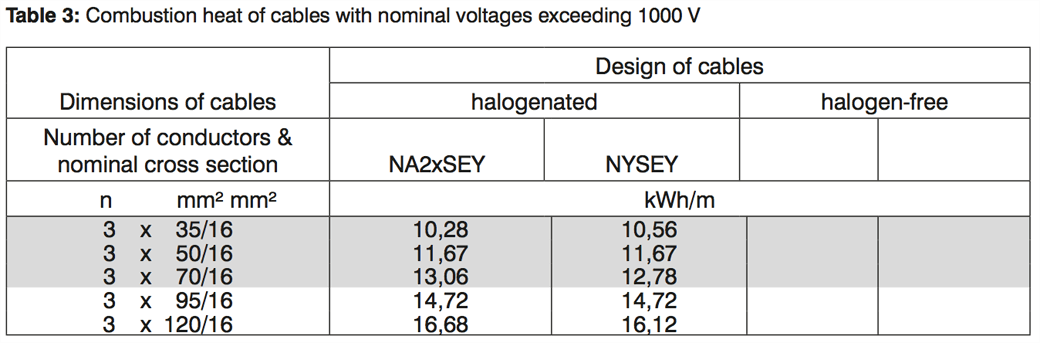
For high voltage cables, there is simply no data available. The planner must calculate it by hand or using a tool like Cableizer.
In order to verify our calculation, we designed a couple of cables listed in the VdS table above. The construction data for N2XSEY cables were taken from the German cable manufacturer Faber Kabel using their data sheet Mittelspannungskabel N2XSEY nach VDE 0276-620.
| Foto from Faber Kabel | 3D preview in Cableizer | 2D preview in Cableizer |
|---|---|---|
 |
 |
 |
The editor automatically calculates weight and the combustion heat. The output is as follows:
| N2XSEY 3x50/16 with datasheet: |
N2XSEY 3x120/16 with datasheet: |
|---|---|
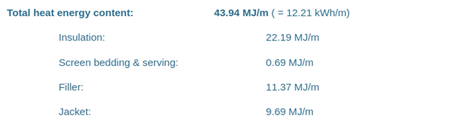 |
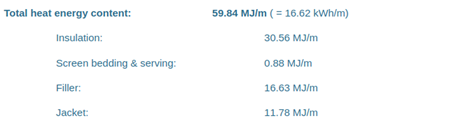 |
Let us compare these values with the values from VdS:
| Cable | Value from VdS | Value from Cableizer | Deviation |
|---|---|---|---|
| N2XSEY 3x50/16 | 11.67 kWh/m | 12.21 kWh/m | +4.6% |
| N2XSEY 3x120/16 | 16.68 kWh/m | 16.62 kWh/m | -0.4% |
Not bad considering the large range of values for the heat of combustion coefficients. This brings us to the next point: Where is the data from?
The sources for the heat of combustion value for non-fuel products are rather difficult to find. So a key task to develop this new feature was the search for the necessary values, if possible from multiple sources in order to gain a higher confidence in the validity of the data.
Our sources were:
Note that we did not systematically take the highest value found but the most plausible value based on multiple sources. And some best-guess estimation was needed for specially treated material like Fibrous polyolefin copolymer.